Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
CAS No.: 110-82-7
Molecular Weight:84.15948
Introduction:
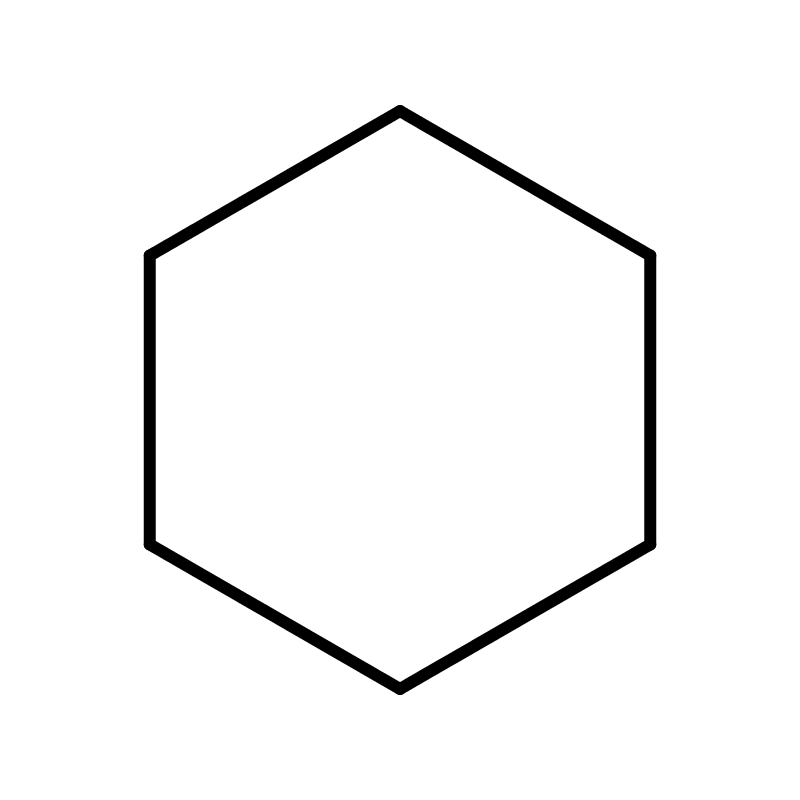
Cyclohexane (C6H12) is an organic compound. At room temperature, cyclohexane appears as a colorless liquid with a mild, sweet odor. It belongs to the chemical class of aliphatic hydrocarbons, specifically a saturated cyclic hydrocarbon. This classification reflects its structure, which consists of a ring of six carbon atoms, each bonded to two hydrogen atoms, forming a stable, non-aromatic configuration.
Contact information
PRODUCT DESCRIPTION
Use and Manufacturing
1 Chemical Reactivity
Reactivity with Water: No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
2 Environmental Fate
Biological. Microbial degradation products reported include cyclohexanol (Dugan, 1972;Verschueren, 1983), 1-oxa-2-oxocycloheptane, 6-hydroxyheptanoate, 6-oxohexanoate, adipic acid,acetyl-CoA, succinyl-CoA (quoted, Verschueren, 1983), and cyclohexanone (Dugan, 1972; Kecket al., 1989). Photolytic. The following rate constants were reported for the reaction of cyclohexane and OHradicals in the atmosphere: 5.38 x 10-12 cm3/molecule?sec at 295 K (Greiner, 1970); 6.7 x 10-12cm3/molecule?sec at 300 K (Darnall et al., 1978); 6.69 x 10-12 cm3/molecule?sec at 298 (DeMoreand Bayes, 1999); 7.0 x 10-12 cm3/molecule?sec (Atkinson et al., 1979); 7.49 x 10-12cm3/molecule?sec (Atkinson, 1990). A photooxidation reaction rate constant of 1.35 x 10-16cm3/molecule?sec was reported for the reaction of cyclohexane with NO3 in the atmosphere(Atkinson, 1991). Chemical/Physical. The gas-phase reaction of cyclohexane with OH radicals in the presence ofnitric oxide yielded cyclohexanone and cyclohexyl nitrate as the major products (Aschmann et al.,1997). Cyclohexane will not hydrolyze because it has no hydrolyzable functional group.
3 Potential ExposureCyclohexane is used as a chemicalintermediate; as a solvent for fats, oils, waxes, resins, cer-tain synthetic rubbers; and as an extractant of essential oilsin the perfume industry
4 Produe MethodCyclohexane is fractionated from crude oil and may bereleased wherever petroleum products are refined, stored,and used. Another large source of general release is inexhaust gases from motor vehicles. It is prepared syntheticallyfrom benzene, by hydrocracking of cyclopentane,or from toluene by simultaneous dealkylation and doublebond hydrogenation.
5 Purification MethodsIt is best to purify it by washing with conc H2SO4 until the washings are colourless, followed by water, aqueous Na2CO3 or 5% NaOH, and again water until neutral. It is then dried with P2O5, Linde type 4A molecular sieves, CaCl2, or MgSO4 then Na and distilled. Cyclohexane has been refluxed with, and distilled from Na, CaH2, LiAlH4 (which also removes peroxides), sodium/potassium alloy, or P2O5. Traces of *benzene can be removed by passage through a column of silica gel that has been freshly heated: this gives material suitable for ultraviolet and infrared spectroscopy. If there is much *benzene in the cyclohexane, most of it can be removed by a preliminary treatment with nitrating acid (a cold mixture of 30mL conc HNO3 and 70mL of conc H2SO4) which converts *benzene into nitrobenzene. The impure cyclohexane and the nitrating acid are placed in an ice bath and stirred vigorously for 15minutes, after which the mixture is allowed to warm to 25o during 1hour. The cyclohexane is decanted, washed several times with 25% NaOH, then water, dried with CaCl2, and distilled from sodium. Carbonyl-containing impurities can be removed as described for chloroform. Other purification procedures include passage through columns of activated alumina and repeated crystallisation by partial freezing. Small quantities may be purified by chromatography on a Dowex 710-Chromosorb W gas-liquid chromatographic column. Flammable liquid. [Sabatier Ind Eng Chem 18 1005 1926, Schefland & Jacobs The Handbook of Organic Solvents (Van Nostrand) p592 1953, Beilstein 5 IV 27.] Rapid purification: Distil, discarding the forerun. Stand distillate over Grade I alumina (5% w/v) or 4A molecular sieves.
Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS