Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
CAS No.: 12125-02-9
Molecular Weight:53.489
Introduction:
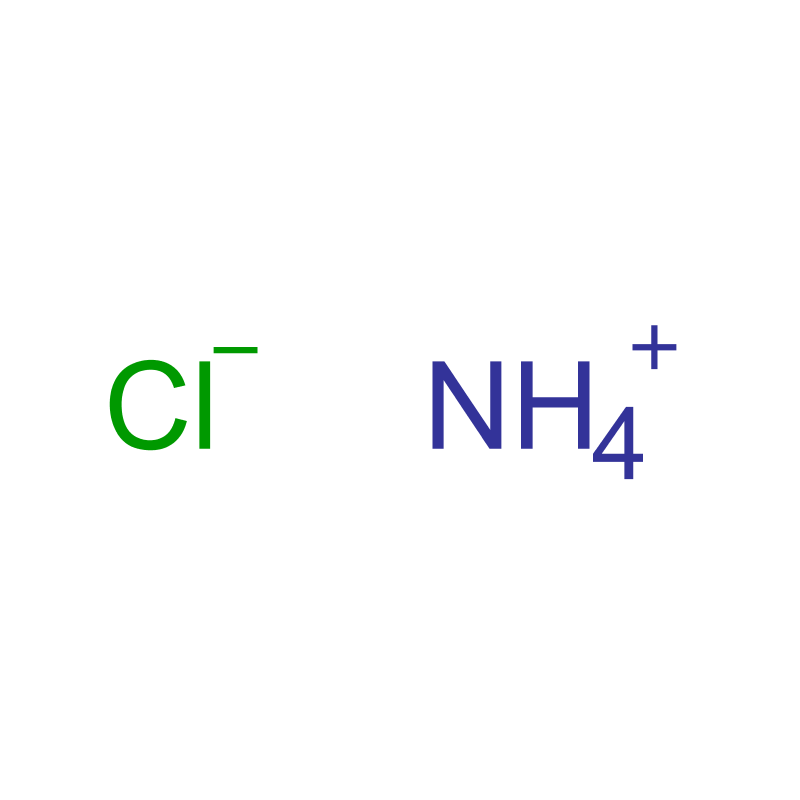
Ammonium chloride (ClH4N) is an inorganic ionic compound. It appears as a white crystalline solid at room temperature, is odorless, and typically forms fine granules or powder. The compound is highly soluble in water, forming a colorless solution, and exhibits hygroscopic properties, meaning it can absorb moisture from the surrounding environment. As an ammonium salt of hydrochloric acid, it belongs to the class of inorganic salts and is widely used in various industrial and scientific applications.
Contact information
PRODUCT DESCRIPTION
Physical and chemical properties
Chemical formula: NH4Cl
Electronic:
Relative molecular mass: 53.49
Character
Colorless crystal or white granular powder, it is a kind of strong electrolyte, which dissolves in water and ionizes to produce ammonium ions and chloride ions. When ammonia gas and hydrogen chloride combine to form ammonium chloride, there will be white smoke. Odorless. The taste is salty, cool and slightly bitter. It has low hygroscopicity, but it can also absorb moisture and agglomerate in wet and rainy weather. Powdered ammonium chloride is very easy to deliquesce, especially qualified products. The moisture absorption point is generally around 76%. When the relative humidity in the air is greater than the moisture absorption point, ammonium chloride will absorb moisture and easily agglomerate. Capable of sublimation (actually the process of decomposition and regeneration of ammonium chloride) without melting point. The relative density is 1.5274. The refractive index is 1.642. Low toxicity, median lethal dose (rat, oral) 1650mg/kg. irritating. Heating to 350 ° C sublimation, the boiling point of 520 ° C.
Soluble in water, slightly soluble in ethanol, soluble in liquid ammonia, insoluble in acetone and ether. Hydrochloric acid and sodium chloride can reduce its solubility in water. Its solubility in water is 29.4g at 0°C, 33.2g at 10°C, 37.2g at 20°C, 41.4g at 30°C, 45.8g at 40°C, and 45.8g at 50°C. 50.4g at 60°C, 55.3g at 60°C, 60.2g at 70°C, 65.6g at 80°C, 71.2g at 90°C, and 77.3g at 100°C). It begins to decompose when heated to 100°C, and can be completely decomposed into ammonia gas and hydrogen chloride gas at 337.8°C. When it is cooled, it will recombine to form ammonium chloride with extremely small particles and appear as white smoke, which is not easy to sink and is extremely difficult to regenerate. Dissolve in water.
The pH of the aqueous solution: Because the ammonium ion ionized in the water is hydrolyzed to make the solution acidic, the pH value of the saturated ammonium chloride solution at room temperature is generally around 5.6. At 25°C, 1% is 5.5, 3% is 5.1, and 10% is 5.0. Ammonium chloride itself can be used as a stable reagent for bismuth and antimony (provide chloride ions for coordination, and its compound and ammonium chloride are co-dissolved in dilute hydrochloric acid when used).
chemical properties
The aqueous solution is weakly acidic, and the acidity increases when heated. It is corrosive to ferrous metals and other metals, especially to copper, and has no corrosive effect on pig iron; when ammonia gas is mixed with hydrogen chloride gas, white smoke will be generated, which is ammonium chloride;
Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS