Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
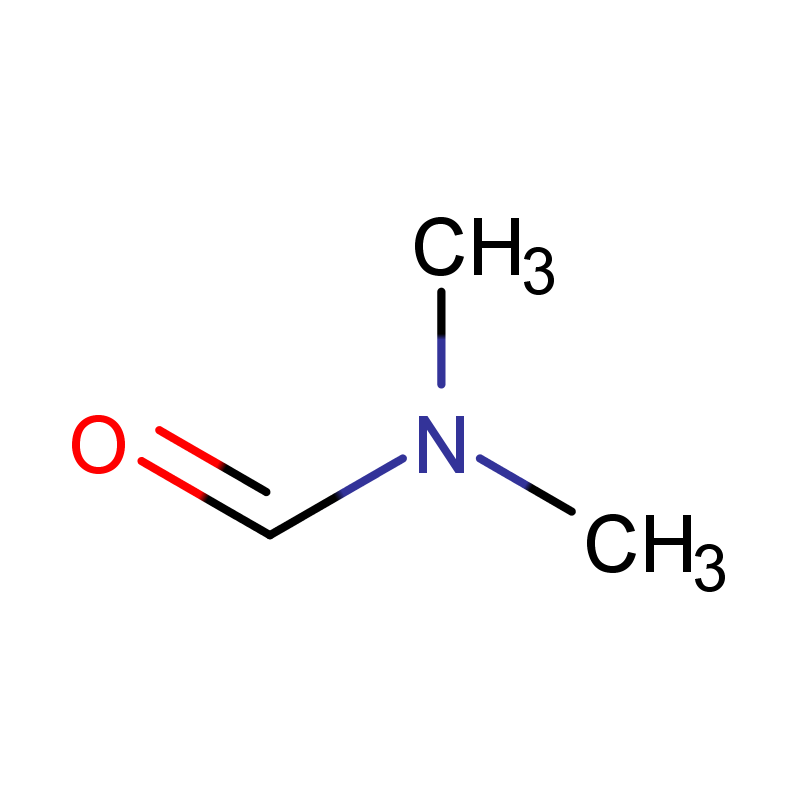
CAS No.: 68-12-2
Molecular Weight:73.09378
Introduction:
N,N-Dimethylformamide (C3H7NO) is a polar aprotic solvent and carboxylic acid derivative, specifically the dimethylamide of formic acid. At room temperature, it is a colorless to pale yellow liquid with a faint, characteristic odor often described as faintly fishy or chloroform-like; it is miscible with water and most common organic solvents.
Contact information
PRODUCT DESCRIPTION
Use and Manufacturing
1 Definition
ChEBI: A member of the class of formamides that is formamide in which the amino hydrogens are replaced by methyl groups.
2 Polymerization
Methylene diisocyanate polymerized violently on contact with dimethyl formamide.
3 Produe Method
Industrial production of N,N-Dimethylformamide (DMF) is via three separate processes (Eberling 1980). Dimethylamine in methanol is reacted with carbon monoxide in the presence of sodium methoxide or metal carbonyls at 110-150°C and high pressure. Alternately, methyl formate is produced from carbon monoxide and methanol under high pressure at 60-100°C in the presence of sodium methoxide. The resulting methyl formate is distilled and then reacted with dimethylamine at 80-100°C and low pressure. The third process involves reaction of carbon dioxide, hydrogen and dimethylamine in the presence of halogen-containing transition metal compounds to yield DMF.
4 Purification Methods
DMF decomposes slightly at its normal boiling point to give small amounts of dimethylamine and carbon monoxide. The decomposition is catalysed by acidic or basic materials, so that even at room temperature DMF is appreciably decomposed if allowed to stand for several hours with solid KOH, NaOH or CaH2. If these reagents are used as dehydrating agents, therefore, they should not be refluxed with the DMF. Use of CaSO4, MgSO4, silica gel or Linde type 4A molecular sieves is preferable, followed by distillation under reduced pressure. This procedure is adequate for most laboratory purposes. Larger amounts of water can be removed by azeotropic distillation with *benzene (10% v/v, previously dried over CaH2), at atmospheric pressure: water and *benzene distil below 80o. The liquid remaining in the distillation flask is further dried by adding MgSO4 (previously ignited overnight at 300-400o) to give 25g/L. After shaking for one day, a further quantity of MgSO4 is added, and the DMF is distillied at 15-20mm pressure through a 3-ft vacuum-jacketed column packed with steel helices. However, MgSO4 is an inefficient drying agent, leaving about 0.01M water in the final DMF. More efficient drying (to around 0.001-0.007M water) is achieved by standing with powdered BaO, followed by decanting before distillation, then with alumina powder (50g/L, previously heated overnight to 500-600o), and distilling from more of the alumina, or by refluxing at 120-140o for 24hours with triphenylchlorosilane (5-10g/L), then distilling at ca 5mm pressure [Thomas & Rochow J Am Chem Soc 79 1843 1957]. Free amine in DMF can be detected by the colour reaction with 1-fluoro-2,4-dinitrobenzene. It has also been purified by drying overnight over KOH pellets and then distilling from BaO through a 10 cm Vigreux column (p 11) [Jasiewicz et al. Exp Cell Res 100 213 1976]. [For efficiency of desiccants in drying dimethylformamide see Burfield & Smithers J Org Chem 43 3966 1978, and for a review on purification, tests of purity and physical properties, see Juillard Pure Appl Chem 49 885 1977.] It has been purified by distilling from K2CO3 under high vacuum and fractionated in an all-glass apparatus. The middle fraction is collected, degassed (seven or eight freeze-thaw cycles) and redistilled under as high a vacuum as possible [Mohammad & Kosower J Am Chem Soc 93 2713 1971]. [Beilstein 4 IV 171.] Rapid purification: Stir over CaH2 (5% w/v) overnight, filter, then distil at 20mmHg. Store the distilled DMF over 3A or 4A molecular sieves. For solid phase synthesis, the DMF used must be of high quality and free from amines.
Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS