Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
CAS No.: 71-23-8
Molecular Weight:60.09502
Introduction:
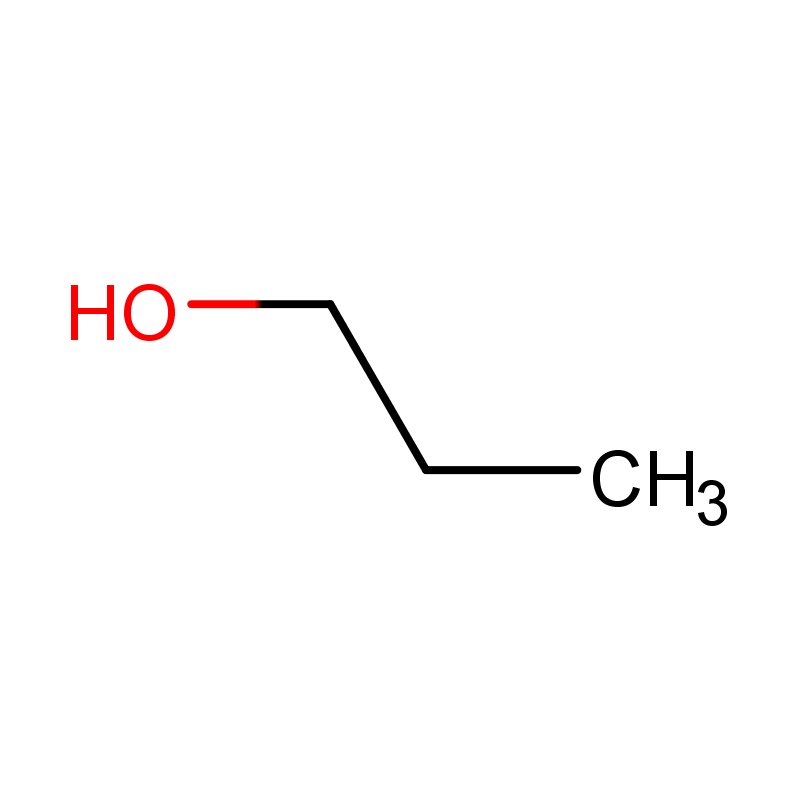
1-Propanol (C₃H₈O) is an organic compound. 1-Propanol (C₃H₈O) is a colorless liquid with a characteristic alcoholic odor at room temperature. It is a primary alcohol, belonging to the broader class of aliphatic alcohols. This compound exhibits moderate volatility and is miscible with water, as well as many organic solvents such as ethanol, acetone, and chloroform.
Contact information
PRODUCT DESCRIPTION
Use and Manufacturing
1 Chemical ReactivityReactivity with Water No reaction; Reactivity with Common Materials: No reactions; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.2 Definitionpropanol: Either of two alcohols with the formula C3H7OH. Propan-1- ol is CH3CH2CH2OH and propan-2-ol is CH3CH(OH)CH3. Both are colourless volatile liquids. Propan-2-ol is used in making propanone (acetone).3 Environmental FateBiological. In activated sludge inoculum, following a 20-d adaptation period, 98.8% CODremoval was achieved. The average rate of biodegradation was 71.0 mg COD/g?h (Pitter, 1976).Using the BOD technique to measure biodegradation, the mean 5-d BOD value (mM BOD/mM 1-propanol) and ThOD were 2.70 and 60.0%, respectively (Vaishnav et al., 1987).Photolytic. Reported rate constants for the reaction of 1-propanol and OH radicals in the atmosphere: 2.3 x 10-12 cm3/molecule?sec at 300 K (Hendry and Kenley, 1979); 2.3 x 10-9L/molecule?sec (second-order) at 292 K (Campbell et al., 1976), 5.33 x 10-12 cm3/molecule?sec at296 K (Overend and Paraskevopoulos, 1978). Based on an atmospheric OH concentration of 1.0 x106 molecule/cm3, the reported half-life of 1-propanol is 1.5 d (Grosjean, 1997).Chemical/Physical. At an influent concentration of 1,000 mg/L, treatment with GAC resulted inan effluent concentration of 811 mg/L. The adsorbability of the carbon used was 38 mg/g carbon(Guisti et al., 1974).4 Potential Exposuren-Propyl alcohol is used as as solventin lacquers, dopes; to make cosmetics; dental lotions; clea-ners, polishes, and pharmaceuticals; as a surgical antiseptic.It is a solvent for vegetable oils, natural gums and resins;rosin, shellac, certain synthetic resins; ethylcellulose, andbutyral; as a degreasing agent; as a chemical intermediate.5 Produe Method1-Propanol is produced commercially by the oxo process byreacting ethylene with carbon monoxide and hydrogen inthe presence of a catalyst to give propionaldehyde, which isthen hydrogenated.6 Purification MethodsThe main impurities in n-propyl alcohol are usually water and 2-propen-1-ol, reflecting the commercial production by hydration of propene. Water can be removed by azeotropic distillation either directly (azeotrope contains 28% water) or by using a ternary system, e.g. by also adding *benzene. Alternatively, for removal of gross amounts of water, reflux over CaO for several hours is desirable, followed by distillation and a further drying. To obtain more nearly anhydrous alcohol, suitable drying agents are firstly NaOH, CaSO4 or K2CO3, then CaH2, aluminium amalgam, magnesium activated with iodine, or a small amount of sodium. Alternatively, the alcohol can be refluxed with n-propylsuccinate or phthalate in a method similar to the one described under EtOH. Allyl alcohol is removed by adding bromine (15mL/L) and then fractionally distilling from a small amount of K2CO3. Propionaldehyde, also formed in the bromination, is removed as the 2,4-dinitrophenylhydrazone. n-Propyl alcohol can be dried down to 20ppm of water by passage through a column of pre-dried molecular sieves (type 3 or 4A, heated for 3hours at 300o) in a current of nitrogen. Distillation from sulfanilic or tartaric acids removes impurities. Albrecht [J Am Chem Soc 82 3813 1960] obtained spectroscopically pure material by heating with charcoal to 50-60o, filtering and adding 2,4-dinitrophenylhydrazine and a few drops of conc H2SO4. After standing for several hours, the mixture is cooled to 0o, filtered and distilled in a vacuum. Gold and Satchell [J Chem Soc 1938 1963] heated n-propyl alcohol with 3-nitrophthalic anhydride at 76-110o for 15hours, then recrystallised the resulting ester from H2O, *benzene/pet ether (b 100-120o)(3:1), and *benzene. The ester was hydrolysed under reflux with aqueous 7.5M NaOH for 45minutes under nitrogen, followed by distillation (also under nitrogen). The fraction with b 87-92o is dried with K2CO3 and stirred under reduced pressure in the dark over 2,4-dinitrophenylhydrazine, then freshly distilled. Also purify it by adding 2g NaBH4 to 1.5L of alcohol, gently flushing with argon and refluxing for 1day at 50o. Then 2g of freshly cut sodium (washed with propanol) is added and refluxed for one day, and finally distilled, taking the middle fraction [Jou & Freeman J Phys Chem 81 909 1977]. [Beilstein 1 IV 1413.]Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS