Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
CAS No.: 71-36-3
Introduction:
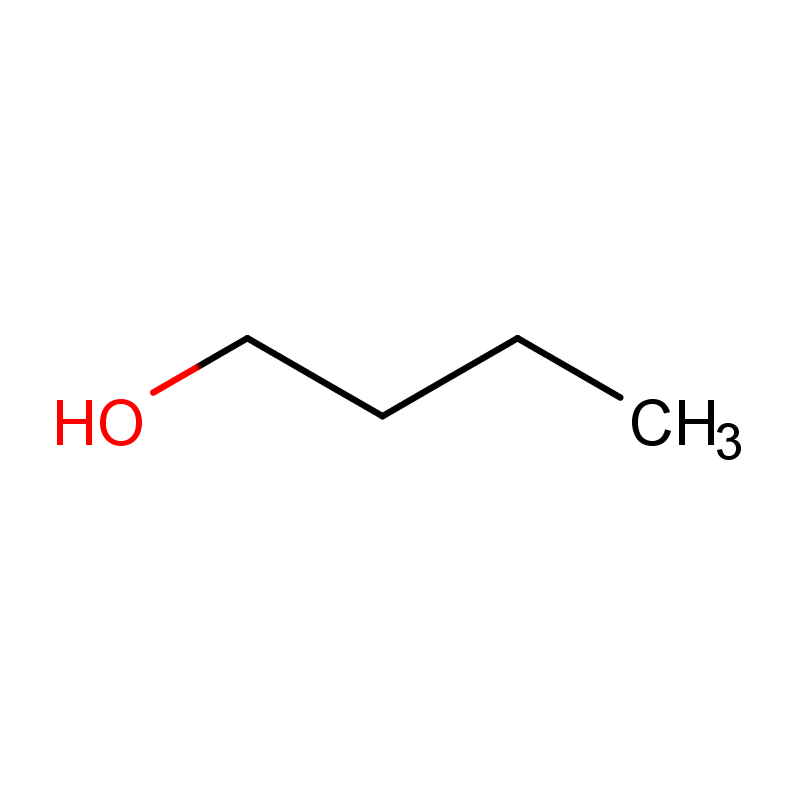
1-Butanol (C4H10O) is a primary alcohol and an organic compound. At room temperature, it exists as a colorless liquid with a mild, characteristic alcoholic odor. It is slightly soluble in water but completely miscible with most common organic solvents such as ethanol, ether, and acetone. The molecule consists of a four-carbon unbranched hydrocarbon chain terminated by a hydroxyl (-OH) functional group, which contributes to its chemical reactivity and physical properties.
Contact information
PRODUCT DESCRIPTION
Use and Manufacturing
1 Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: No reactions; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
2 Definition
Two alcohols that arederived from butane: the primary alcoholbutan-1-ol (CH3(CH2)2CH2OH) and thesecondary alcohol butan-2-ol(CH3CH(OH)CH2CH3). Both are colorlessvolatile liquids used as solvents.
3 General Description
Colorless liquid. Used in organic chemical synthesis, plasticizers, detergents, etc.
4 Methods of Manufacturing
Fermentation method uses corn starch (wheat, corn, etc.) starch as raw material, add water to prepare mash, inoculate acetone butanol bacteria after cooking and sterilize, and ferment at 36~37℃.?The fermentation mash is separated by distillation to obtain butanol, acetone and ethanol, butanol accounts for 55% to 58%, acetone accounts for 30Z to 34%, and ethanol accounts for 7% to 14%.?Molasses or potato starch can also be used as raw materials.?The carbonyl synthesis method, or REPPE method, uses propylene, carbon monoxide and water as raw materials to directly synthesize butanol in one step.?When the reaction temperature is 100~104℃ and the pressure is 1.47MPa, the anionic complex formed by n-butylpyrrolidine, iron pentacarbonyl and water is used as the catalyst component, butanol is used as the solvent, and the conversion rate is 8%~ 10%, the yield of n-butanol is 87%.?The raw materials of this method are easily available and the process is simple, but the conversion rate in one pass is not high, which requires high concentration of carbon monoxide in the raw materials.?In aldol condensation method, acetaldehyde is first added to 10% dilute alkaline solution, and the reaction is performed below 120°C for 0.5 to 2 hours to generate 2-hydroxybutyraldehyde.?When the reaction reaches 50%, the alkali is neutralized, and unreacted acetaldehyde is recovered by distillation, and 2-hydroxybutyraldehyde is obtained from the bottom of the distillation tower.?It is then dehydrated with sulfuric acid or acetic acid to obtain crotonaldehyde.?Using nickel chromium as the catalyst, under the conditions of 180°C and 0.29MPa, the crotonaldehyde is hydrogenated and reduced to butanol, and the reaction mixture is fractionated to obtain the product.?This method has a long process route and serious equipment corrosion, and currently only a few manufacturers use it.
Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS