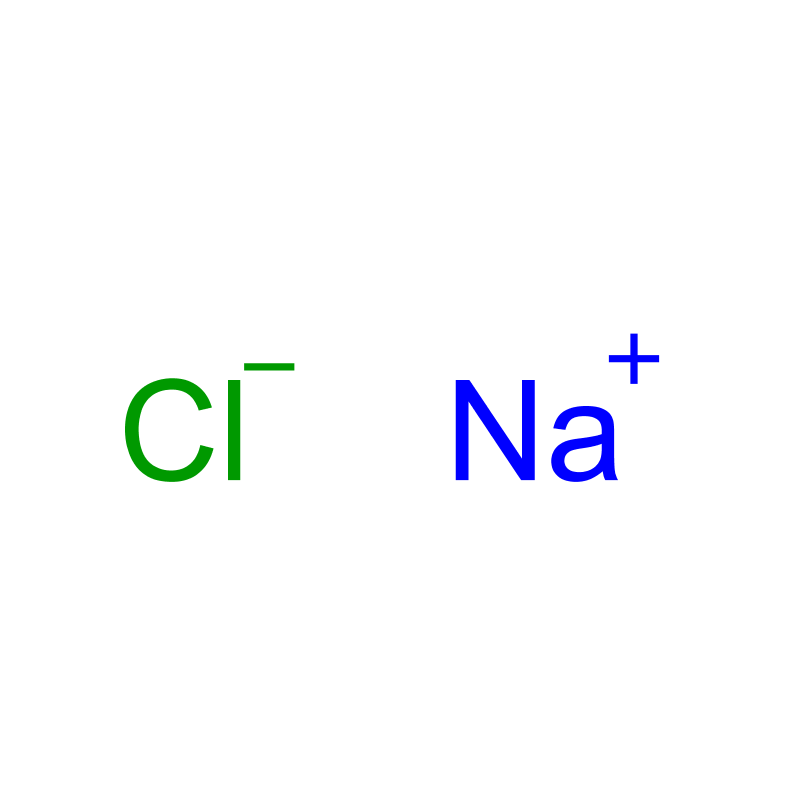Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
CAS No.: 7647-14-5
Molecular Weight:58.44277
Sodium chloride (NaCl) is an inorganic ionic compound composed of sodium and chlorine ions in a 1:1 ratio. At room temperature, it exists as a white crystalline solid with a cubic crystal lattice structure and is typically odorless. Due to the strong electrostatic forces between its constituent ions, sodium chloride exhibits high melting and boiling points, making it stable under ambient conditions. It is highly soluble in water and other polar solvents, where it dissociates into Na⁺ and Cl⁻ ions, contributing to its widespread use in various applications.
Contact information
PRODUCT DESCRIPTION
1.1 NameSodium chloride
1.2 Synonyms
BICINE HIGH PURITY GRADEBORIC ACID SODIUM DECAHYDRATE NF GRADEEINECS 231-598-3MFCD00003477sodium hydrochloride
1.3 CAS No.7647-14-5
1.4 CID5234
1.5 EINECS(EC#)231-598-3
1.6 Molecular FormulaNaCl (isomer)
1.7 InchiInChI=1S/ClH.Na/h1H;/q;+1/p-1
1.8 InChIkeyFAPWRFPIFSIZLT-UHFFFAOYSA-M
1.9 Canonical Smiles[Na+].[Cl-]
1.10 Isomers Smiles[Na+].[Cl-]
2. Properties
2.1 Density1.199 g/mL at 20 °C
2.1 Melting point801 °C(lit.)
2.1 Boiling point100 °C750 mm Hg
2.1 Refractive indexn20/D 1.378
2.1 Flash Point1413°C
2.1 Vapour Pressure1 mm Hg ( 865 °C)
2.2 Precise Quality57.95860
2.2 PSA0.00000
2.2 SolubilityH2O: soluble
2.3 ViscosityViscosity of saturated aqueous solution = 1.93 mPa-s
2.4 Appearancecolourless crystals or white powder
2.5 StorageAqueous sodium chloride solutions are stable but may cause theseparation of glass particles from certain types of glass containers.Aqueous solutions may be sterilized by autoclaving or filtration.The solid material is stable and should be stored in a well-closedcontainer, in a cool, dry place.It has been shown that the compaction characteristics and themechanical properties of tablets are influenced by the relativehumidity of the storage conditions under which sodium chloridewas kept.
2.6 Chemical PropertiesSodium chloride, NaCl, also known as common salt and halite, is a white crystalline solid.It is soluble in water,slightly soluble in alcohol, and melts at 804 °C (1480 °F). Sodium chloride is the most important sodium mineral and occurs naturally in seawater, underground deposits, and brine wells.Sodiumchlorideis a basic raw material for the production of chlorine,sodium hypochlorite, sodium bisulfate,soda ash, and hydrogen chloride. Sodium chloride is also used in food preparation, fertilizers, and by highway departments to control icy road conditions.
2.7 Physical PropertiesSodium chloride is the familiar compound commonly referred to as salt or table salt. Th emineral form of sodium chloride is halite and is found in natural deposits throughout theworld. It accounts for approximately 2.7% by weight of the dissolved minerals in seawater.Sodium chloride is an ionic compound existing as a white crystalline cubic structure of alternatingsodium and chloride ions. Sodium chloride is essential for life, with the average adultrequiring about 1 to 2 grams per day. Salt supplies sodium and provides numerous essentialfunctions such as maintaining water balance in cells, taking part in nerve signal transmissionand muscle contraction.
2.8 Color/FormWhite
2.9 CorrosivitySodium chloride solutions are corrosive to base metals
2.10 DecompositionWhen heated to decomposition it emits toxic fumes of /hydrochloric acid and disodium oxide/.
2.11 PH5.5-6.5(1 tablet in 100 mL purified water)
2.12 Water Solubility360 g/L (20 oC)
2.13 Spectral PropertiesIndex of refraction: 1.5442
2.14 StabilityStable. Incompatible with strong oxidizing agents.
2.15 StorageTemp+15C to +30C
2.16 Surface Tension110 mN/m at 850 deg C /Molten sodium chloride/
3. Use and Manufacturing
3.1 Agricultural UsesHalite is a naturally occurring sodium chloride (NaCl) deposit. The most abundant potash mineral deposit is sylvite (KCl). Sylvite with halite forms the common potash ore, called sylvinite.
3.2 DefinitionChEBI: An inorganic chloride salt having sodium(1+) as the counterion.
3.3 General DescriptionA white crystalline solid. Commercial grade usually contains some chlorides of calcium and magnesium which absorb moisture and cause caking.
3.4 Produe MethodSodium chloride occurs naturally as the mineral halite. Commercially,it is obtained by the solar evaporation of sea water, by mining,or by the evaporation of brine from underground salt deposits.
3.5 Purification MethodsIt is recrystallised from a saturated aqueous solution (2.7mL/g) by passing in HCl gas, or by adding EtOH or acetone. It can be freed from bromide and iodide impurities by adding chlorine water to an aqueous solution and boiling it for some time to expel free bromine and iodine. Traces of iron can be removed by prolonged boiling of solid NaCl in 6M HCl; the crystals are then washed with EtOH and dried at ca 100o. Sodium chloride has been purified by sublimation in a stream of pre-purified N2 and collected by electrostatic discharge [Ross & Winkler J Am Chem Soc 76 2637 1954]. For use as a primary analytical standard, analytical reagent grade NaCl should be finely ground, dried in an electric furnace at 500-600o in a platinum crucible, and allowed to cool in a desiccator. For most purposes, however, drying at 110-120o is satisfactory.
3.6 UsageFacilitates the cross-coupling of organostannanes with iodides without using palladium.
Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS