Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
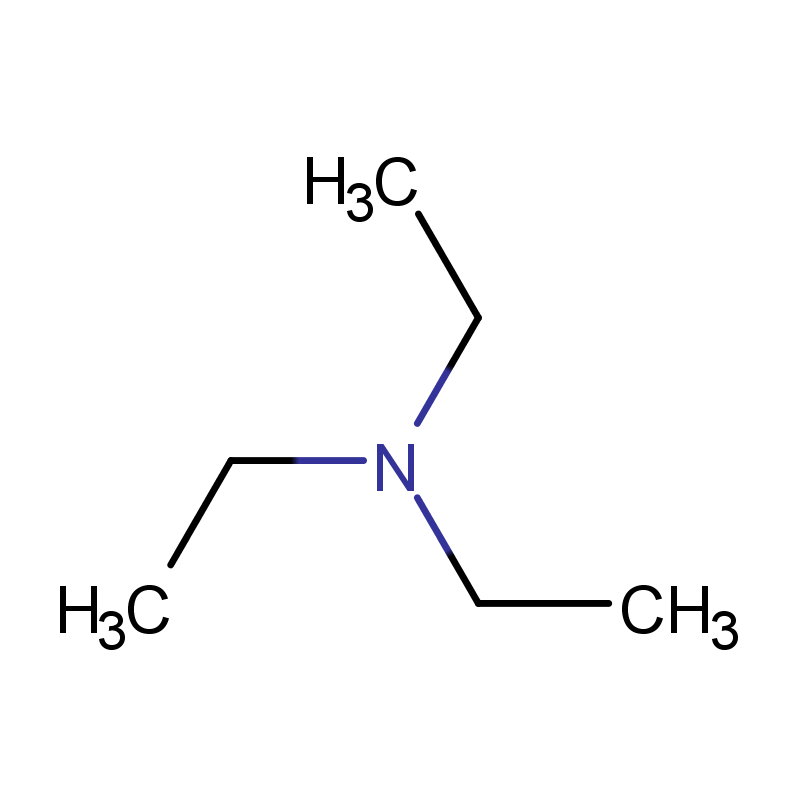
CAS No.: 121-44-8
Molecular Weight:101.19000
Introduction:
Triethylamine (C6H15N) is an organic compound. At room temperature, triethylamine appears as a colorless liquid with a strong, fishy odor. This compound belongs to the class of tertiary amines, which are derivatives of ammonia where all three hydrogen atoms have been replaced by alkyl groups. Specifically, in the case of triethylamine, these alkyl groups are ethyl groups. As such, it exhibits characteristic properties of amines, including basicity and nucleophilicity, making it an important reagent in various chemical processes.
Contact information
PRODUCT DESCRIPTION
Use and Manufacturing
1 DefinitionChEBI: A tertiary amine that is ammonia in which each hydrogen atom is substituted by an ethyl group.2 Environmental FatePhotolytic. Low et al. (1991) reported that the photooxidation of aqueous tertiary aminesolutions by UV light in the presence of titanium dioxide resulted in the formation of ammoniumand nitrate ions.Chemical/Physical. Triethylamine reacted with NOx in the dark to form diethylnitrosamine. Inan outdoor chamber, photooxidation by natural sunlight yielded the following products:diethylnitramine, diethylformamide, diethylacetamide, ethylacetamide, diethylhydroxylamine,ozone, acetaldehyde, and peroxyacetyl nitrate (Pitts et al., 1978).3 Methods of Manufacturing1. Available with ethanol and ammonia methy. The ethanol and liquid ammonia are preheated in a preheater (150 ± 5 ℃) in the presence of hydrogen gas and then fed into a first reactor (190 ± 2 ℃) equipped with a copper-nickel-clay catalyst and The second reactor (165 ± 2 ℃) synthesis, resulting in a ethylamine; diethylamine; triethylamine mixture, after condensation, and then by ethanol spray absorption triethanolamine crude, the final separation; dehydration and fractionation , Collecting 88-90 ℃ distillate triethylamine. (99%) 150 standard cubic meters, t; hydrogen gas (99%), the standard consumption of raw materials: ethanol (95%) 2300kg, t; Refining method: often contain ethylamine, diethylamine and acetaldehyde and other impurities, generally refined by fractional distillation. To remove traces of primary and secondary amines, acetic anhydride, benzoic anhydride or phthalic anhydride may be added and refluxed after fractionation. The distillate may be dried with activated alumina or solid potassium hydroxide and then distilled. Other purification methods are: the triethylamine into hydrochloride, and then ethanol recrystallization, to the melting point of 254 ℃, with sodium hydroxide aqueous solution to free the triethylamine, the solid potassium hydroxide after drying, In the presence of sodium in the nitrogen stream was distilled pure.2. In the presence of oxygen, ethanol and ammonia in copper, nickel, clay catalyst in the presence of heating synthesis reaction, generating one, Di- and triethylamine mixtures. The mixture is condensed, extracted and absorbed to obtain crude product. Crude and then separated, standing layered, dehydration, distillation that was the product.
3. Mix ethanol (kg), liquid ammonia (kg) and hydrogen (standard m3) in a proportion of 23:50:15 and then preheat to 145-155 ℃, then feed the first reactor with copper nickel clay catalyst (About 190 ℃) and the second reactor (about 165 ℃) to form a mixture of ethylamine, diethylamine and triethylamine, condensed and condensed, collecting 88 ~ 90 ℃ distillate triethylamine.
4. Synthesis: Ethyl chloride and ammonia from heating under pressure derived.
Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS