Products
Our products cover multiple core areas such as fine chemicals, basic chemicals, and specialty chemicals, and are widely used in key industries such as coatings, building materials, pharmaceuticals, new energy, and new materials.
CAS No.: 14484-69-6
Molecular Weight:142.07
Introduction:
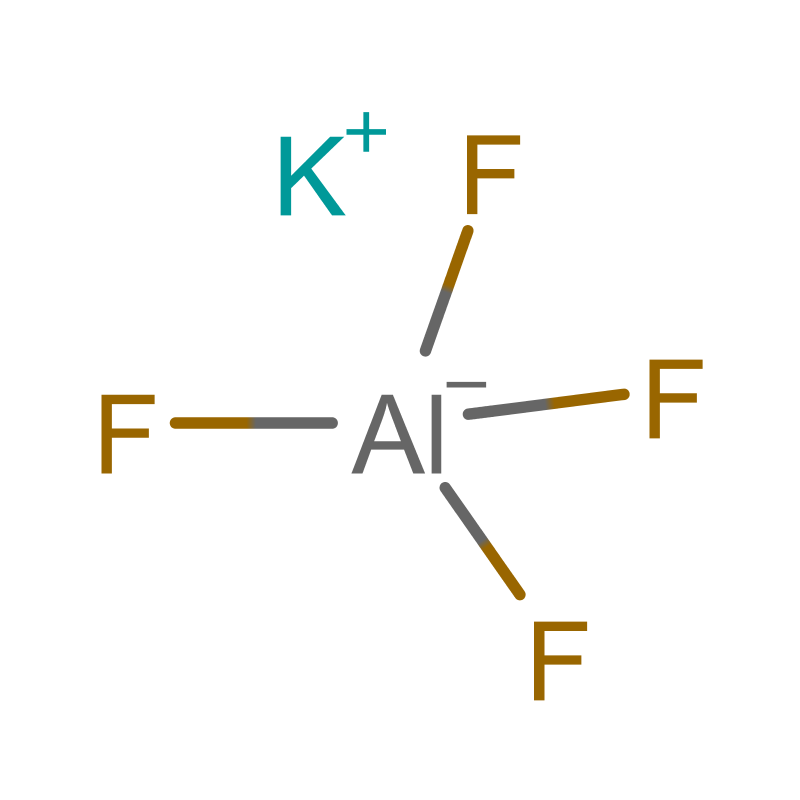
Potassium Aluminum Fluoride (AlF4K) is an inorganic coordination compound that belongs to the class of alkali metal aluminum fluorides. At room temperature, it exists as a white crystalline solid with no characteristic odor. The compound is generally stable under ambient conditions and is often encountered in powder or granular form, depending on its method of synthesis and intended application. Its structure typically features aluminum in a tetrahedral coordination environment surrounded by fluoride ions, with potassium ions serving as counterions to maintain charge neutrality.
Contact information
PRODUCT DESCRIPTION
Potassium aluminum fluoride was first synthesized in the early 20th century as part of studies on molten salt systems and their applications in metallurgy. It was likely prepared through the reaction of aluminum fluoride with potassium fluoride under controlled heating conditions. This synthesis typically occurs in aqueous or molten environments, where stoichiometric ratios of the reactants are carefully maintained to ensure complete formation of the desired compound. Potassium aluminum fluoride finds significant use in the aluminum smelting industry, where it serves as a fluxing agent to lower the melting point of electrolytic bath components in the Hall–Héroult process. It is also employed in the production of specialty glasses, ceramics, and optical materials due to its unique ionic conductivity and thermal stability. Additionally, it has niche applications in welding fluxes and brazing compounds. While not commonly found in biological systems, the compound can be synthesized from naturally occurring fluoride minerals such as cryolite (Na3AlF6) through ion-exchange reactions with potassium salts. Industrially, it is often prepared by reacting potassium fluoride with aluminum fluoride hexahydrate at elevated temperatures, followed by calcination to remove residual moisture and achieve the desired crystallinity.
Potassium aluminum fluoride is considered to be moderately toxic upon ingestion or inhalation, with potential for both acute and chronic health effects. Exposure may lead to irritation of the respiratory tract, coughing, and shortness of breath, while skin or eye contact can result in irritation or chemical burns. Long-term exposure to aluminum-containing fluorides may contribute to skeletal fluorosis or other systemic effects related to aluminum accumulation. The compound is not currently classified as a carcinogen by the International Agency for Research on Cancer (IARC) or the Environmental Protection Agency (EPA), though prolonged exposure should be minimized to prevent potential health risks. For safe handling and storage, potassium aluminum fluoride should be kept in tightly sealed containers in a cool, dry, and well-ventilated area. It should be isolated from strong acids, particularly hydrofluoric acid, which can release toxic fluoride and hydrogen fluoride vapors upon reaction. Storage areas should also be free from incompatible materials such as alkali metals or oxidizing agents to prevent hazardous interactions.
Factory Showcase
Certificate of Honor



Fast transport time
Inventory 2-3 working days New manufacturing 7-10 working days
FAQ
Q:What about your price?
A:Our price is very competitive because we are a factory. Feel free to contact us if you are interested in our products.
Q:Can I go to your factory to visit?
A:Of course, we welcome customers from all over the world to visit our factory.
Q:How about the delivery time?
A:Within 3-15 days after we confirm you requirement.
Q:What is your terms of payment ?
A:T/T, L/C at sight, Cash, Western Union are all accepted if you have other payment,please contact me.
Q: Do you provide samples?
A:Yes,we could offer the sample for you to test but please pay freight fee.
INQUIRY
Application scenarios
![Industrial & Heavy Manufacturing Industrial & Heavy Manufacturing]() VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.
VIEW MOREIndustrial & Heavy ManufacturingThis article explores key products such as Soda Ash, Silicon Dioxide, Cryolite, Potassium Tetrafluoroaluminate, Industrial Sodium Chloride, Water Reducer, Dimethyl Sulfoxide (DMSO), and DMF in typical use cases.![Food & Nutritional Health Food & Nutritional Health]() VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.
VIEW MOREFood & Nutritional HealthThis article details specific applications of our product lines including Citric Acid, L-Sodium Glutamate, Sweeteners, NMN, L-Ergothioneine, Tocopherol (Vitamin E) , Carboxymethyl Cellulose (CMC) , as well as Methionine and Lysine in food and animal nutrition.![Pharmaceuticals & Personal Care Pharmaceuticals & Personal Care]() VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.
VIEW MOREPharmaceuticals & Personal CareThis article systematically analyzes the application value of our core products including L-Tartaric Acid, Cholesterol, Creatine Phosphate, Sodium Hyaluronate, Kojic Acid, Sodium Lauroyl Glutamate, and Cetearyl Alcohol in real-world scenarios.![Agriculture & Ecological Treatment Agriculture & Ecological Treatment]() VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.
VIEW MOREAgriculture & Ecological TreatmentThis article details the practical applications of our products including Ammonium Chloride, Ammonium Bicarbonate, Hinokitiol, EDTA series (EDTA, EDTA-2Na, EDTA-4Na), and Calcium Chloride in agricultural productivity, green plant protection, and ecological water treatment.![High-Performance Materials & Polymers High-Performance Materials & Polymers]() VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.
VIEW MOREHigh-Performance Materials & PolymersThis article focuses on core raw materials in our product line, including 2,5-Furandicarboxylic Acid (FDCA), Erucic Acid, Hydroxypropyl Methacrylate (HPMA), 2-Ethyl-4-methylimidazole, Hydroquinone, and Acetic Anhydride, analyzing their key roles in polymer synthesis, modification, and processing.![General Chemicals & Fine Synthesis General Chemicals & Fine Synthesis]() VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
VIEW MOREGeneral Chemicals & Fine SynthesisThis article systematically introduces the core raw materials in our General Chemicals & Fine Synthesis product line—Acetic Anhydride, Benzyl Chloride, Dimethylamine Aqueous Solution, Creatinine, Creatinine Hydrochloride, Calcium Stearate, Magnesium Stearate, EDTA-2Na/4Na—in practical applications within laboratories, CRO/CDMOs, and small-to-medium-sized factories.
RELATED PRODUCTS